Classification of Salts











Classification of Salts
Classification of Salts:
The different types of salts are: normal salt, acid salt, basic salt and double salt.
1. Neutral Salt: A salt that does not contain any replaceable hydrogen atoms or hydroxyl groups is called a normal salt. Example: Na2 SO4 obtained in the reaction between and NaOH is a normal salt because it is formed by the complete replacement of both the H atoms of
. Similarly, calcium sulphate
sodium phosphate (
) and potassium phosphate (
) are also normal salts.
2. Acid Salt: When an acid is not completely neutralized by a base, the salt produced will contain replaceable hydrogen atoms. Hence, it may further take part in the reaction with the base as an acid. Such a salt is called an acid salt. For example, the salt produced in the reaction between NaOH and
is an acid salt because it is capable of further reaction with the base NaOH to produce the normal salt
.
Thus, an acid salt may be defined as follows: A salt that contains replaceable hydrogen atoms is called an acid salt. Examples: and
are examples of acid salts.
3. Basic salt: When a base reacts with lesser amount of acid than, is necessary for complete neutralization, the salt produced contain hydroxyl group(s)(OH) also. Such a salt is called a basic salt.
Example: Pb(OH)Cl. These salts are called basic salts, because they can further react with the acids to form and the corresponding normal salts. Thus, a basic salt is formed when a base reacts with a lesser amount of an acid than is necessary for the formation of a normal salt
.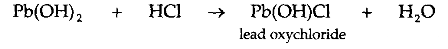
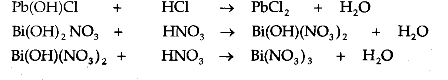
Double salt: In a double salt, there are two different negative ions and/or positive ions. For example, the mineral dolomite, ,
, contains both
and
ions. Hence, it is a double salt. Potash alum:
, also is a double salt. Double salts exist only in the solid state. When dissolved in water, they break up into a mixture of two separate salts. For example, when potash alum is dissolved in water, it breaks up as follows.
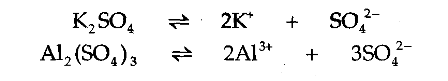
'Alum' is an example of ______________ | |||
| Right Option : B | |||
| View Explanation | |||
Which of the following are correct ? (a) Salts formed by the reaction of strong acid with a strong base are called neutral salts. (b) Salts formed by the reaction of a weak acid with a strong base are called basic salts. | |||
| Right Option : C | |||
| View Explanation | |||
Salts formed by reaction of a strong acid and weak base are called _________________________ . | |||
| Right Option : B | |||
| View Explanation | |||
Students / Parents Reviews [10]
A marvelous experience with Abhyas. I am glad to share that my ward has achieved more than enough at the Ambala ABHYAS centre. Years have passed on and more and more he has gained. May the centre flourish and develop day by day by the grace of God.

Archit Segal
7thBeing a parent, I saw my daughter improvement in her studies by seeing a good result in all day to day compititive exam TMO, NSO, IEO etc and as well as studies. I have got a fruitful result from my daughter.

Prisha Gupta
8thMy experience with Abhyas is very good. I have learnt many things here like vedic maths and reasoning also. Teachers here first take our doubts and then there are assignments to verify our weak points.

Shivam Rana
7thI have spent a wonderful time in Abhyas academy. It has made my reasoning more apt, English more stronger and Maths an interesting subject for me. It has given me a habbit of self studying

Yatharthi Sharma
10thAbhyas Methodology is very good. It is based on according to student and each child manages accordingly to its properly. Methodology has improved the abilities of students to shine them in future.

Manish Kumar
10thAbout Abhyas metholodology the teachers are very nice and hardworking toward students.The Centre Head Mrs Anu Sethi is also a brilliant teacher.Abhyas has taught me how to overcome problems and has always taken my doubts and suppoeted me.

Shreya Shrivastava
8thMy experience with Abhyas academy is very good. I did not think that my every subject coming here will be so strong. The main thing is that the online tests had made me learn here more things.

Hiya Gupta
8thIt was good as the experience because as we had come here we had been improved in a such envirnment created here.Extra is taught which is beneficial for future.

Eshan Arora
8thIt has a great methodology. Students here can get analysis to their test quickly.We can learn easily through PPTs and the testing methods are good. We know that where we have to practice

Barkha Arora
10thIt was a good experience with Abhyas Academy. I even faced problems in starting but slowly and steadily overcomed. Especially reasoning classes helped me a lot.
